AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Nuclear charge of carbon11/10/2023 
It is worth noting that the arrangement of electrons in different 2 p orbitals, necessitated by Hund’s rule, produces a configuration of lower energy. In such a case it is assumed that the reader knows that the two 2 p electrons are not spin paired. To date, about 115 different elements have been discovered by definition, each is chemically unique.\) may also be found. 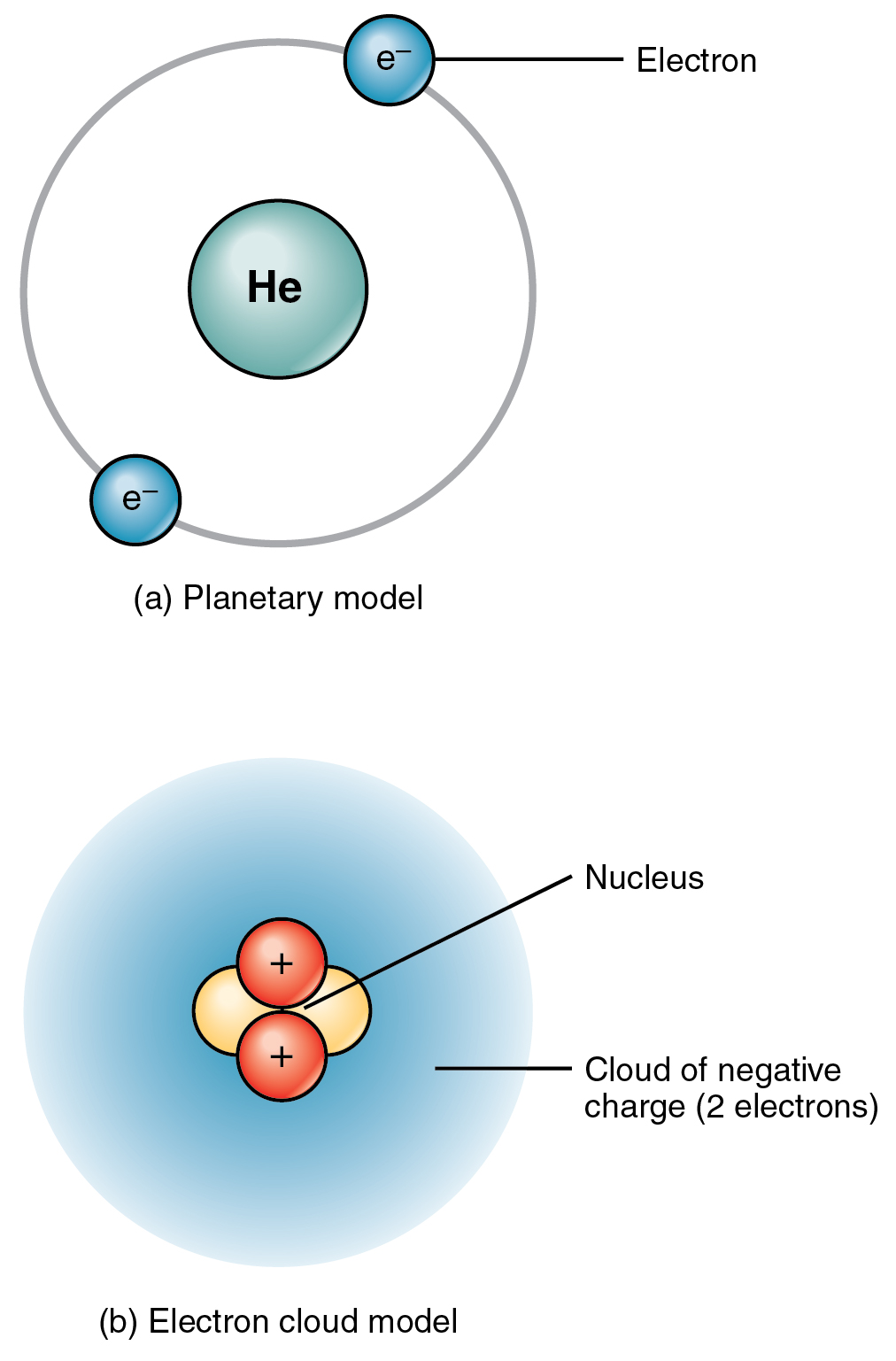 
The chemical name of the element and its symbol are uniquely tied to the atomic number thus the symbol "Sr" stands for strontium, whose atoms all have Z = 38. 
You can think of the atomic number as a kind of serial number of an element, commencing at 1 for hydrogen and increasing by one for each successive element. Moseley found that the square roots of these frequencies give a straight line when plotted against Z this enabled him to sort the elements in order of increasing atomic number. The frequencies of these X-rays are unique to each element, and they increase uniformly in successive elements. Carbon-14 emits beta particles as it decays and reverts back to nitrogen. He found this in a class of X-rays emitted by an element when it is bombarded with electrons. Carbon C-14 is an unstable isotope of carbon created when a neutron collides with a nitrogen atom, causing capture of the neutron and release of the proton converting nitrogen to a carbon with fourteen nucleons (6 protons and 8 neutrons). Moseley searched for a measurable property of each element that increases linearly with atomic number. In the neutral atom, the Z protons within the nucleus are balanced by Z electrons outside it.Ītomic numbers were first worked out in 1913 by Henry Moseley, a young member of Rutherford's research group in Manchester. Each proton carries an electric charge of +1, so the atomic number also specifies the electric charge of the nucleus. It is, rather, the number of protons in the nucleus, which we call the atomic number and denote by the symbol Z. What single parameter uniquely characterizes the atom of a given element? It is not the atom's relative mass, as we will see in the section on isotopes below. The resulting electrically-charged atom is called an ion. Most nuclei contain roughly equal numbers of neutrons and protons, so we can say that these two particles together account for almost all the mass of the atom.īecause the electrons of an atom are in contact with the outside world, it is possible for one or more electrons to be lost, or some new ones to be added. Its mass is almost the same as that of the proton. As its name implies, this particle carries no electrical charge. The other nuclear particle is the neutron. This means that in any atom, the number of protons in the nucleus (often referred to as the nuclear charge) is balanced by the same number of electrons outside the nucleus. Protons are the carriers of positive electric charge in the nucleus the proton charge is exactly the same as the electron charge, but of opposite sign. The nucleus is itself composed of two kinds of particles. The radius of an atom must be defined arbitrarily, such as the boundary in which the electron can be found with 95% probability. The latter has no definite outer boundary, so neither does the atom. It is common (but somewhat misleading) to describe the volume of space in which the electrons of an atom have a significant probability of being found as the electron cloud. The extremely small mass of the electron (1/1840 th the mass of the hydrogen nucleus) causes it to behave as a quantum particle, which means that its location at any moment cannot be specified the best we can do is describe its behavior in terms of the probability of its manifesting itself at any point in space. Almost all of the volume of an atom consists of empty space in which electrons, the fundamental carriers of negative electric charge, reside.Almost all of the mass of an atom is contained within a tiny (and therefore extremely dense) nucleus which carries a positive electric charge whose value identifies each element and is known as the atomic number of the element.The most notable of these achievements was Ernest Rutherford's famous 1911 alpha-ray scattering experiment, which established that: The precise physical nature of atoms finally emerged from a series of elegant experiments carried out between 18.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
